“Wildlife health issues are not just a biodiversity concern. The emergence of infectious diseases and antimicrobial resistance in wildlife must be viewed through a multi-disciplinary lens that captures issues and threats not only in people and domesticated animals but also the wider environment. One Health is the only approach to mitigating health risks in today’s world.”

I am APHA’s lead scientist for Wildlife, based at the National Wildlife Management Centre. Our mission is to provide evidence-based, impartial advice and operational services on preventing, predicting and mitigating wildlife disease and human-wildlife conflicts in support of Defra and the devolved governments. Our science is directed at solving practical problems to enable flourishing wildlife and native species and to safeguard animal health. Let me share with you some examples of the One Health approach being central to my team’s work.
What is worse than eating an apple and finding a worm?
Finding worm eggs in fox poo…
Echinococcus multilocularis, a parasitic tapeworm present in Europe, is the causative agent of human alveolar echinococcosis, a potentially fatal infection of the liver. The life cycle of the tapeworm is quite complex and involves, in the UK, the red fox (the definitive host that harbours adult worms) and various species of voles (the intermediate host in which the worm eggs develop). Humans accidently become infected through the ingestion of food or water contaminated with the eggs of the parasite. The United Kingdom currently has E. multilocularis free status, and as such, must implement a surveillance programme to provide evidence for the absence of the tapeworm. Previous research from the Wildlife team estimated the fox population size at around 230,000 individuals, unevenly distributed throughout the country.
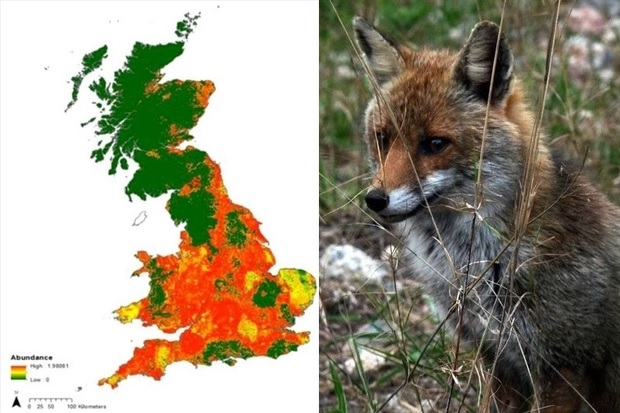
As the national reference laboratory for Echinococcus, we process around 380 fox faecal samples every year from various sources. We use molecular techniques to look for traces of tapeworm egg DNA in the faeces.
The possibility of introduction of E. multilocularis in UK wildlife exists, and is most likely to be via an imported or returning dog that has not had any worming treatment. The UK has therefore adopted preventive measures and requires tapeworm treatment for all dogs entering the UK to reduce the risk.
The establishment of the disease in wildlife is currently assessed by Defra as low but uncertain. Should one of the UK’s safeguards against the tapeworm fail, we are developing mathematical models to predict the probability and cost of successfully eliminating a localised outbreak of the tapeworm based on different control programmes. We are also investigating through these models other possible introduction pathways, for example through beavers imported from infected areas in Europe as part of reintroduction projects.
African swine fever…the heat is on

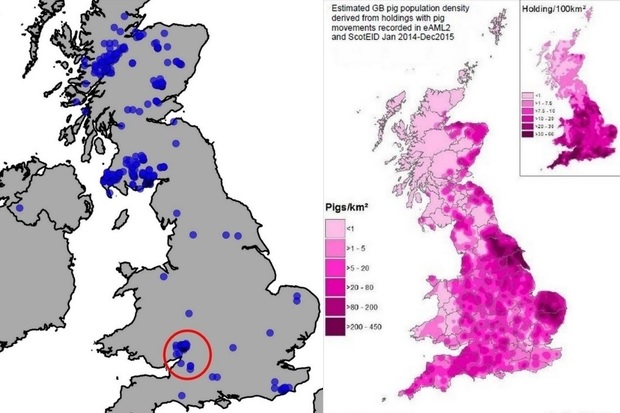
In the UK, native wild boars were hunted to extinction in the 13th century but several free-living populations have become established in the last few decades as the result of escapes from commercial farms. Wild boar are adapted to living almost everywhere, from woodlands to city centres, and are very successful at reproducing! Each female can produce up to two litters of three to seven piglets per year. This has resulted in their range (the area they can be found) and population size rapidly expanding in the UK, and more generally throughout the northern hemisphere.
Feral wild boar may present a serious epidemiological risk to neighbouring livestock as they can carry and transmit a number of important livestock diseases. We recently built a mathematical model to explore whether the current population of feral wild boar in the Forest of Dean, which has one of the largest populations of wild boar in the UK, can act as a reservoir for exotic (not usually found in the UK) or notifiable diseases such as foot-and-mouth (FMD) or African swine fever (ASF). A (wildlife) reservoir is a population in which a pathogen can be permanently maintained and from which infection is transmitted to a target population (e.g. a livestock population) in which we would need to control the disease. We concluded that while the current population of boar is insufficient to maintain FMD, if boar are allowed to spread beyond their current range into adjacent areas, they might become a self-sustaining reservoir. The Wildlife team is expanding on this work to investigate under what conditions ASF could spread in this same wild boar population and to predict persistence of the virus under different wild boar population control levels.
ASF is a rather hot topic at the moment, if you excuse the pun. It is a highly contagious disease affecting both domestic and wild pigs. While African swine fever is not a risk to human health, it is responsible for serious production and economic losses in pig production systems and cases must be reported to the World Organisation for Animal Health (OIE). The virus continues to spread outside of Africa, and as of February 28th 2020, 22 countries notified new or ongoing outbreaks including 11 in Europe.
Because of our geography (wild boars are not good swimmers!), ASF would most likely enter the UK through the import of contaminated pork products so this risk is currently set to “medium”. Our wildlife modellers are busy assessing the risks of ASF transmission from wild boar to domestic pigs in the UK, a crucial step in ensuring government policies are proportionate to exclude the disease as well as ensuring a rapid and effective contingency response if necessary.
-----------
Since 2009, surveillance for wildlife disease in GB has been the responsibility of the Great Britain Wildlife Disease Surveillance Partnership, under the chair of the APHA’s Diseases of Wildlife Scheme (DoWS). Paul Duff, APHA’s wildlife expert group veterinary lead, relates an unusual investigation into antimicrobial resistant bacteria making the jump from humans to some of our sea mammals.
Seals and humans - sharing more than just the beach
As part of a continuing investigation, APHA has identified the bacterial species Klebsiella pneumoniae subspecies pneumoniae (Kpp) in free-living common seal pups on the east coast of England. The bacteria, of suspected human origin, possess extensive antimicrobial resistance and caused disease in the pups.
Antimicrobial resistance (AMR) is the ability of a microorganism (like bacteria) to stop a medicine (such as antibiotics) from working against it. As a result, standard treatments become ineffective, infections persist and may spread to others.
More recently, Kpp was isolated again, in a stranded common seal pup this time, and more worryingly, the bacteria possessed resistance to a critically important antimicrobial, as classified by the World Health Organisation. Critically important antimicrobials are those used for treating human infections with multi-drug resistant bacteria, resistant to other therapeutic options. Klebsiella pneumoniae (Kpp) is the second most frequent cause of Gram-negative bacteraemia (bacteria which are good at hiding from our immune system and are often resistant to antibiotics) and urinary tract infections in humans!

We believe that multi-drug resistant bacterial pathogens are contaminating the coastal environment and resulting in a small number of opportunistic, life threatening, infections in juvenile seals. The most likely route of coastal contamination is through the discharge from neighbouring sewage outflows.
When APHA DoWS started this work examining clinical samples from the RSPCA wildlife unit which specialises in the care of stranded seals, we did not suspect that wild seals would be infected with bacteria with AMR, simply because these wild seals would not have been exposed to such bacteria in the marine environment and had not been treated with antibiotics!
There is growing scientific evidence that wildlife throughout the world may be infected with bacteria which possess extensive antimicrobial resistance. It is often assumed that these bacteria originate from contact with anthropogenic (human) sources such as farms and human waste that pollute the environment with AMR bacteria and/or with antimicrobials. However, it is becoming clearer that assigning the source and directionality of AMR dissemination is challenging and that more novel approaches focusing on the human-animal-environmental interface is required.
This investigation work involved collaboration between the specialised RSPCA seal rescue centre, where examination and sampling of the seals occurred, the surveillance input of the Animal and Plant Health Agency (APHA) Diseases of Wildlife Scheme, the Bacteriology department at APHA Weybridge, where molecular examinations were undertaken and input from colleagues at Public Health England (PHE).
Find out more
Enjoyed reading this blog? You may be interested in reading some of the other wildlife blogs we have published.
Be sure never to miss new stories from the APHA Science Blog Team – subscribe here for instant email alerts.
Did you know, we are also active on social media? Follow us on Twitter (@APHAgovuk), Facebook (APHAgov) and LinkedIn.

Recent Comments